The dietary supplement is referred to as Health Functional Food (HFF) in South Korea, which is a category with massive potential as reported by Korea Functional Food Material Information System [1] who reported that the import value had been increasing from 2014 to 2018 with a YOY of 10.5%. During Covid-19, the demand amongst Korean consumers for health functional foods blew up in popularity. According to Statista's report, the sales of health functional products skyrocketed to 579% from January 27 to February 11, 2020 [2] (See Figure 1). The sales of vitamin products grew over 2,000% compared to the previous year.

(Figure 1. Sales of functional products from January 27 to February 11, source: Statista Research Department)
HFF is food manufactured with functional raw materials or ingredients. From the definition of HFF, we can tell the ingredient compliance is on the front burner for the manufacturing the HFF products. However, the requirements of HFF ingredients are complicated. So to help enterprises transcend the regulatory obstacles, this article will focus on a comprehensive introduction to the South Korean HFF ingredients.
1. Mandatory Requirements
Superordinate laws: Health Functional Food Act [3] Enforcement Decree of Health Functional Food Act [4] Enforcement Rule of Health Functional Food Act [5]
Supporting regulations: Health Functional Food Code [6] Regulation on Certification of Functional Ingredients and Specification [7] |
Above are the laws and requirements of HFF in South Korea, amongst which the Health Functional Food Code [6] belongs to, though it's a supporting regulation, it is the primary standard for entities for reference. It details the standards and specifications for manufacturing, testing, importing, storing, and selling HFF in Korea. Manufacturing that follows the requirements stated in Health Functional Food Code [6], enterprises can sell or import HFF without a pre-evaluation. (S. Korea Health Functional Food Code contains standards and specifications of different ingredients. No official English version has been published so far but ChemLinked provides translation services, and you can choose to translate only the parts needed. Please contact us via food@chemlinked.com.)
Health Functional Food Act [3], along with its enforcement decree [4] and enforcement rule [5], regulates the overarching policy of HFF, such as the supervision department, the subordinate regulations, etc. The Regulation on Certification of Functional Ingredients and Specification [7] specifies the certificate requirements. The Labeling Standard of Health Functional Food [8] states the provisions about labelling an HFF product.
2. Functional Materials
As stipulated in Article 2 of Health Functional Food Code [6], an HFF product contains functional materials, general food materials, and food additives:
No. | Category | Note |
1 | Functional Materials (including noticed and individually recognized materials):
| Shall comply with Health Functional Food Code [6] |
2 | General Food Materials | Shall comply with Food Code [9] |
3 | Food Additives | Shall comply with Food Additive Code [10] |
2.1 Noticed Ingredients
As previously mentioned, when manufacturing products with the notified functional materials following the manufacture process and methods under the Health Functional Food Code [6], the enterprise could sell or import the products in or to South Korea directly. MFDS has notified twenty-eight nutrient ingredients and sixty-eight functional raw materials so far.
It's highly recommended that overseas enterprises confirm the ingredient list prior to exporting dietary supplement business to S. Korea.(Find the whole list at ChemLinked Translation: South Korean Health Functional Food Ingredients [11]).
2.2. Individually Recognized Ingredients
To produce or import HFF with “non-notified ingredients”, the entities shall apply for a certificate for the effectiveness and safety of the relevant functional material. Until now, MFDS has reviewed and published more than 200 kinds of individually recognized ingredients.
Note that only the applicant can use the relevant recognized ingredients.
2.2.1 Application for Individually Recognized Ingredients Certificate
Following Regulation on Certification of Functional Ingredients and Specification [7], the manufacturer or importer shall submit the following four application materials to MFDS to evaluate the effectiveness and safety of the relevant ingredient:
(1) Application letter, including the company name, major business, product information, and other necessary information of the enterprise.
(2) Dossiers:
A summary of the dossier
Information about the ingredient's origin, development process, certification, and the using status
Manufacture method
Property of the ingredient
Specification, test method, and test result of the active ingredient
Specification and test method of hazard ingredients
Safety information
Effectiveness information
Acceptable daily intake, precautions, etc.
(3) Samples
(4) Inspection report issued by the inspection and testing institutions designated by MFDS.
2.2.2 Application Procedure
Having received the application materials, MFDS will conduct a review and consult with the Health Functional Food Deliberation Committee (HFF Committee). When the safety and effectiveness are confirmed, MFDS will release the approval to the individually recognized ingredient (See Figure 2). The result will also be unveiled on MFDS website.
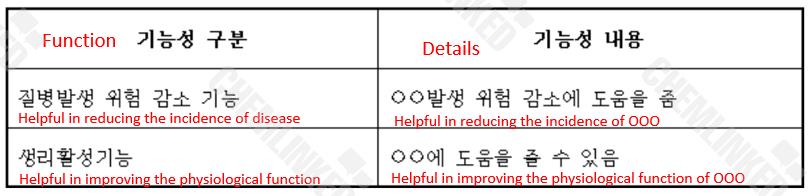
(Figure 2. Example of Approved Function of Individually Recognized Raw Material)
MFDS can request the applicant to complement the data when further information is in need. The applicant needs to submit complementary documents otherwise, the application for "individually recognized ingredient" would be rejected.
 (Figure 3. Evaluation Procedure)
(Figure 3. Evaluation Procedure)
The official charge for certificating “individual recognized ingredient” is 1,900,000 KRW (about 1,710 USD). The period costs 120 days. It might take longer if the documents are incomplete.
2.3 Transforming Individually Recognized Ingredients to Noticed Ingredients
After being approved as “individually recognized ingredient” for six years and used as a functional ingredient in over 50 products, the relevant ingredient would be added into Health Functional Food Code [6].
By adding “individually recognized ingredients” into Health Functional Food Code [6], the Government aims to enlarge the choice of available functional ingredients that would benefit the dietary supplement industry.
3. Health Functional Food Tendency
3.1 Customized Health Functional Food
MFDS launched a trial run on “Customized Health Functional Food System” on April 27, 2020. In the official notification, MFDS allowed seven firms to engage in a pilot program involving formulation of the “Customized Health Functional Foods”.
The "Customized Health Functional Food” is functional food formulated with a mix of different active ingredients to meet consumer’s individual nutritional or physiological requirements. This product should be made based on the recommendations of an expert consultant or genetic testing.
South Korea is expected to see a booming demand for functional raw materials as the new business of HFF has been launched. Although at present, overseas enterprises are ineligible to participate in the "Customized Health Functional Food" business, it is still an opportunity for suppliers to export ingredients or raw materials to South Korea. The COVID-19 pandemic has also expedited a surge in need of functional food active ingredients. (Find more at South Korea’s Customized Health Functional Food System [12])
3.2 Function Claim Scheme for General Foods
On July 15, 2020, during the 10th FOODPOLIS International Conference, the MFDS introduced the "function claim scheme for general foods" aiming to expand the scale of the industry.
This new scheme was made to allow general food to label “function claims”. Nevertheless, to avoid the abuse of function claims, MFDS would only permit the claims when the health effects of the foods had been proved with ample scientific evidence. Additionally, MFDS proposed twenty-nine raw materials applicable for general foods with function claims as well as the requirements of manufacture, labelling, and advertising.
MFDS was planning to release the exposure draft in August this year. Unfortunately, as the COVID 19 situation deteriorated in South Korea, the promulgation of the new scheme was delayed. Once it’s been implemented, many firms are expecting to benefit from this new requirement. (Find more at South Korea Introduces the Function Claim Scheme for General Foods in the 10th FOODPOLIS International Conference [13])
More Information about Health Functional Food





 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









