On May 30, 2019, State Administration for Market Regulation (SAMR) opened the public consultation period on the proposed cancellation of 104 regulatory support documents[1] currently used by authorities to aid in regulation of various sectors of the market. Alteration and/or extension of the registration certificates of health food, infant formula and food for special medical purposes could be influenced if the proposed changes enter into effect. Details are:
| S/N | Supporting document proposed to be canceled | Purpose | Regulatory basis |
| 28 | Document verified by provincial food and drug administration on the production and sales of health food when the registration license is valid | Registration extension of health food | Administrative Measures for Registration and Filing of Health Foods |
| 29 | Proof of changing manufacturer name and/or address | Registration alteration of health food | |
| 30 | Proof of changing items displayed in registration certificate and its attachments | Registration alteration of FSMP | |
| 31 | Registration certificate and its attachments | Registration alteration and extension of infant formula | Administrative Measures for Registration of Infant and Young Children Milk Powder Formula Recipes |
| 32 | Proof of changing manufacturer name, address name, and legal representative | Registration alteration of infant formula |
Deadline for comments is June 29, 2019. You may submit feedback by:
Visiting China legislative information website (http://zqyj.chinalaw.gov.cn/index), see image below.

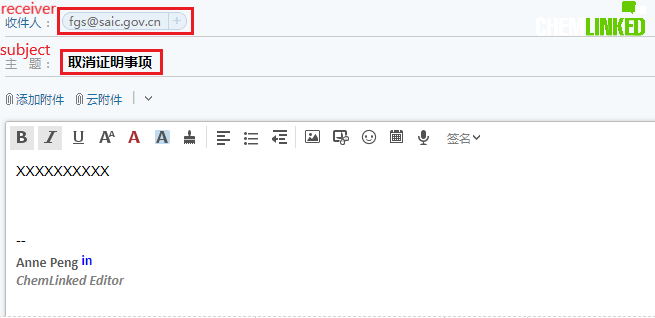
Or sending email to [email protected]. Subject of the email should be “取消证明事项” (items to be cancelled)


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









