On Nov. 6th, MFDS released a guideline [1] introducing a new consultation service to help enterprises expedite approval of functional foods. On Nov. 13th, NIFDS (The National Institute of Food and Drug Safety Evaluation) released a brief report [2] detailing the implementation procedures. Here’re the content.
Necessity of introducing a consultation service
In South Korea, it has generally taken the government around 120 days to assess functional food ingredients and issue results (this step excludes time required to conduct safety review, etc.). However, if the scientific evidence supporting the application is deemed insufficient during the review the application is rejected, requiring applicants to start the whole process again. This results in significant time and resources wasted for both the applicant and the government.
Procedures for implementing the new consultation mechanism
Preparation stage:
Consultation stage:
Processing stage:
(View more at the picture below)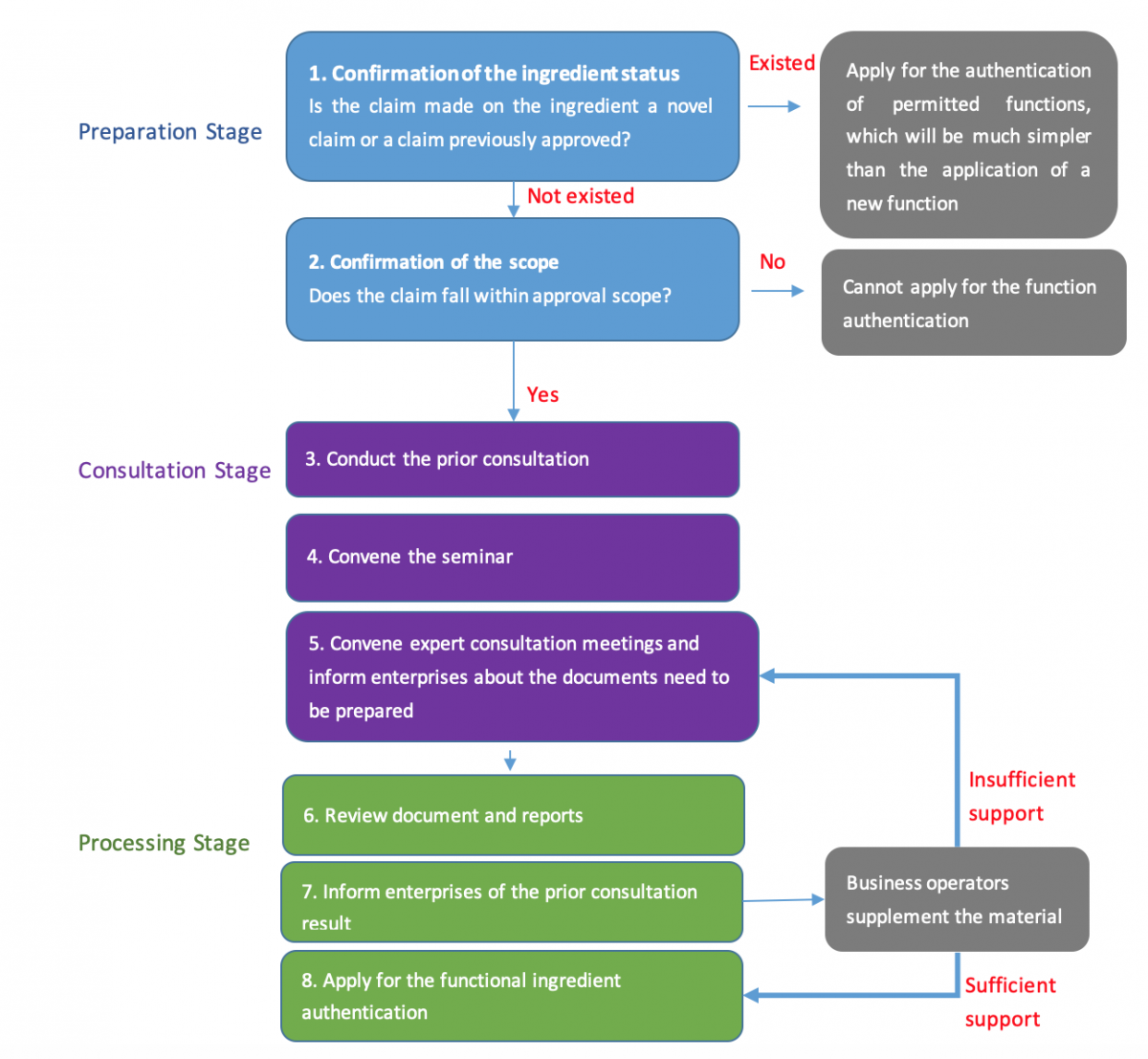


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









