In 2017, China implemented a dual track system to regulate the market access of imported health food. The system allows imported products to enter the Chinese market by filing or registration with SAMR. Since then, every year dozens of imported products entered the market successfully. However, due to strict barriers, the registration approval for imported health food became stagnant just one year after its implementation. As a consequence, product filing becomes the only way that actually works for imported products when exporting to China.
1. Overview of Product Filing/Registration Performance

Imported Health Food Obtained Filing Approval in H1 2022
Product Name | Filing no. | Country of origin |
Jamieson Multivitamin | 食健备J202200000001 | Canada |
Healthy Care Zinc Vitamin C Strawberry Flavor Chewable Tablets | 食健备J202200000002 | Australia |
As mentioned earlier, only 17 imported products obtained registration approvals back in 2017 and no more approvals since then.
On the contrary, a total of 164 imported health food have granted filing approvals by the end of H1 2022. Compared with the filing data by the end of H1 2021, it is revealed that 23 imported health food has been granted filing approvals from SAMR over the past 12 months. Notably, only two products obtained filing approvals in 2022 so far. However, it doesn’t necessarily mean that 2022 will be a tough year for product filing. Coincidentally, in H1 2021, only two imported products were approved for filing as well, while H2 2021 witnessed 21 products were granted filing approvals. Therefore, it is very likely to see more imported products to gain filing approval in the next few months.
2. Product Filing Data from H2 2021 – H1 2022
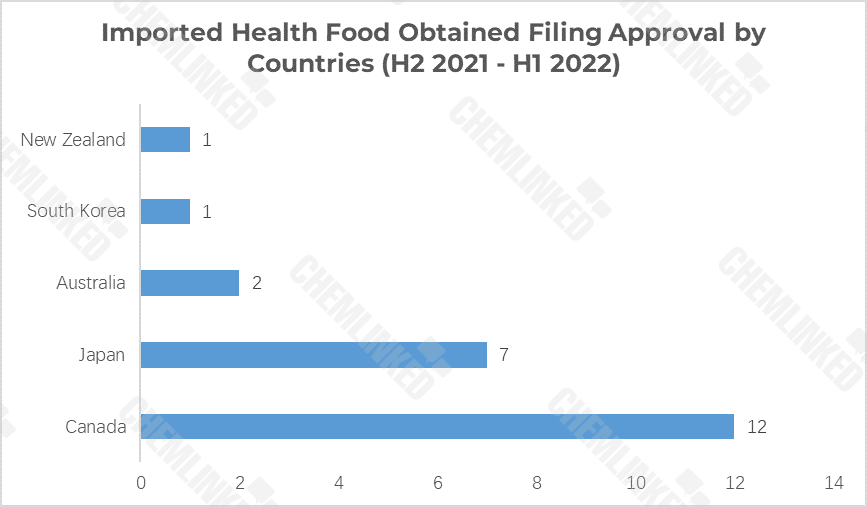 Over the past 12 months, a total of 23 imported health food from 5 countries were granted filing approvals. Among them, 12 are from 2 Canadian brands, namely, Jamieson and Kirkland. Japanese brand I.B gained 7 filing approvals, followed by 2 approvals from Australia’s AusNatures and Nature’s Care. South Korea’s NutrionLife and New Zealand’s Good Health each snatched 1 approval for their products.
Over the past 12 months, a total of 23 imported health food from 5 countries were granted filing approvals. Among them, 12 are from 2 Canadian brands, namely, Jamieson and Kirkland. Japanese brand I.B gained 7 filing approvals, followed by 2 approvals from Australia’s AusNatures and Nature’s Care. South Korea’s NutrionLife and New Zealand’s Good Health each snatched 1 approval for their products.
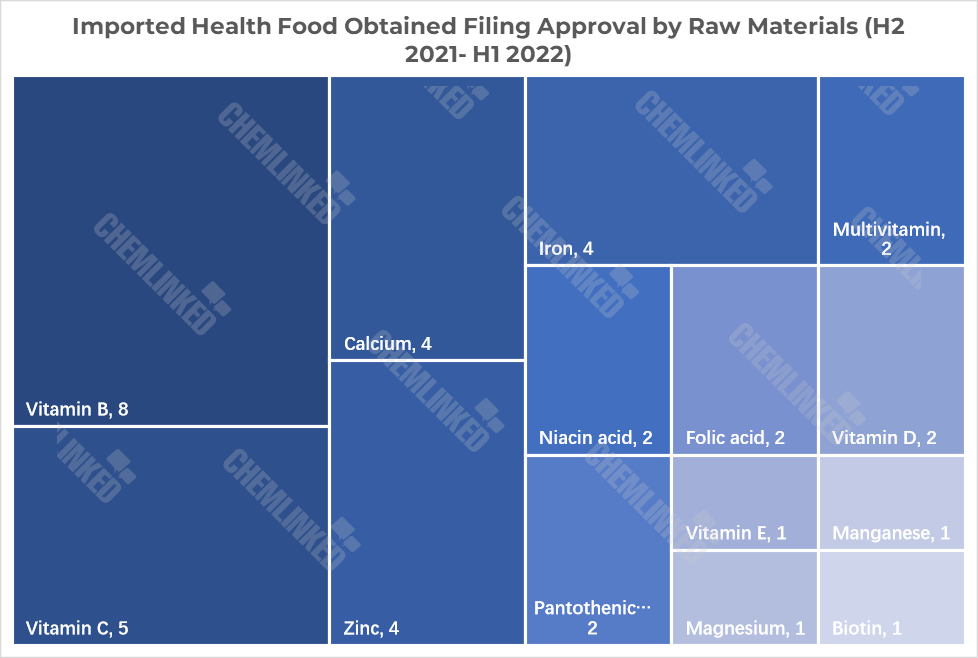 Notes: A health food product is allowed to contain one, two or more raw materials. For example, a product from Good Health contains four raw materials including magnesium, manganese, vitamin D and vitamin B6.
Notes: A health food product is allowed to contain one, two or more raw materials. For example, a product from Good Health contains four raw materials including magnesium, manganese, vitamin D and vitamin B6.
China adopts a positive list for raw materials used for health food subject to filing. As shown in the chart above, 14 kinds of nutrients were used in the 23 products. Vitamin B was the most popular nutrient, which was widely used in 8 products. The least frequently used nutrients were vitamin E, manganese, magnesium and biotin.
3. Supervision Trend on the Market Access of Imported Health Food
Despite registration approval for imported health food has been in a standstill for many years, a series of competent authorities’ recent actions seem as endeavors to lower the barrier of filing, so as to promote the development of the health food industry. As mentioned earlier, one of the prerequisites for products to be eligible for filing is to use the raw materials on the positive list. It’s obviously delighted to see the list is probably to be longer in the near future, which will bring more opportunities to both Chinese domestic and overseas health food manufacturers and brands:
In December 2021, SAMR released the draft of Health Food Raw Materials Directory - Nutrient Supplement (2022) and Health Function Catalogue Allowed for Health Food Claims - Nutrient Supplement (2022). China plans to add four food ingredients into the raw material directory for nutrient supplement, including new sources of calcium, folic acid and DHA. Since DHA was listed in this directory, "supplement n-3 polyunsaturated fatty acids" was added as a new health function for nutrient supplement correspondingly. Once the two drafts are approved, it means imported products can use these four ingredients and the new claim for health food filing. More details.
Probiotic health foods are currently subject to the supervision of registration. However, on the 6th China Special Food Conference held in October 2021, it was revealed that SAMR was considering the feasibility to push forward the filing of some probiotic health food products. Specifically, for probiotic health foods that intend to claim the common function of probiotics like “regulating intestinal flora”, they may obtain the product certificate via filing in the future. More details.
In June 2022, Chinese Nutrition Society announced to seek technical materials such as relevant standards and testing report about three raw materials, namely, Lycopene, Phytosterol and Inulin. This action was interpreted as a clear signal that these three raw materials may be allowed to be used in health food for filing soon.
With the regulatory landscape turning more friendly, the springtime for imported health product filing is just around the corner. ChemLinked and its founder, REACH24H, have jointly launched the health food filing service to help overseas health food & supplement brands seize more opportunities. Feel free to contact us.
Feel free to contact ChemLinked if you need the imported health food filing data in details
Recommended reading:


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










