Unlike China and some other countries, Japan’s has a wider scope of what is considered a health food and this is reflected in the diversified array of products on offer. According to a research organization headquartered in Japan Fuji Keizai Group, market volume of “health & beauty foods” (H&B foods) is expanding and the market value was estimated at 1400 billion JPY in 2017[1]. Notably, green tea and drinkable dairy products account for 90% of the market.
 * “H&B foods” is a concept of a prepackaged food intended for people in a healthy/semi-healthy/mild disease state to consume in order to maintain/promote/recover health and beauty. (Source: public speech)
* “H&B foods” is a concept of a prepackaged food intended for people in a healthy/semi-healthy/mild disease state to consume in order to maintain/promote/recover health and beauty. (Source: public speech)
Foods with Health Claims
We can attribute part of the prosperity of the health food industry in Japan to its evolving regulatory system. From a global perspective the term health food/functional food is an ambiguous term which varies based on the regulatory frameworks adopted in different regions. In Japan health foods are defined as foods with health claims[2]. Food with health claims are a distinctive category which differ from general foods and pharmaceutical products and can be considered a borderline product that overlaps between these two discrete classifiers possessing characteristics of both. In Japan there are 3 types of health food. Although products from all the three categories are allowed to use health claims on labeling and advertising, the application procedure and regulatory compliance requirements varies considerably.
Food with health claims are a distinctive category which differ from general foods and pharmaceutical products and can be considered a borderline product that overlaps between these two discrete classifiers possessing characteristics of both. In Japan there are 3 types of health food. Although products from all the three categories are allowed to use health claims on labeling and advertising, the application procedure and regulatory compliance requirements varies considerably.
ØFoods for special health uses (FOSHU, TOKUHO)
TOKUHO are scientifically recognized as helpful for maintaining and promoting health and are permitted to bear claims such as "Slows cholesterol absorption." They are analogous to Chinese health foods (保健食品) which bear the “blue hat” certification mark, in that both are used to positively impact risk factors associated with specific chronic diseases and/or improve specific physiological functions. The use of any claims must be substantiated using scientific and clinical data using rigorous assessment standards and must be reviewed and approved by regulatory authorities prior to market access. No claims implying medical therapeutic function in the treatment of disease can be used. In Japan the relevant competent authority evaluates the product from both an efficacy and safety perspective, and the Secretary-General of the Consumer Affairs Agency gives approval for the labelling of each food product that meets regulatory requirements. Similar to China a specific certification mark is used. (see picture below). The scheme was established in 1991, and licenses have been granted to over 1,000 products as of Jan. 10, 2019. Approval requires considerable financial investment and is time consuming as clinical trials are mandatory.
The scheme was established in 1991, and licenses have been granted to over 1,000 products as of Jan. 10, 2019. Approval requires considerable financial investment and is time consuming as clinical trials are mandatory.
ØFoods with nutrient function claims (FNFC)
FNFC can be used to supplement and help meet daily micronutrient requirements (vitamins, minerals, etc.). The use of nutrient claims are based on well-established scientific consensus. The use of these claims is constrained to a limited number of nutrients and associated claims which are well defined within Japanese national standards. Products which fulfill these requirements can use functional claims (labeling and advertising) without notifying the relevant authorities. This scheme was launched in 2001.
ØFoods with function claims
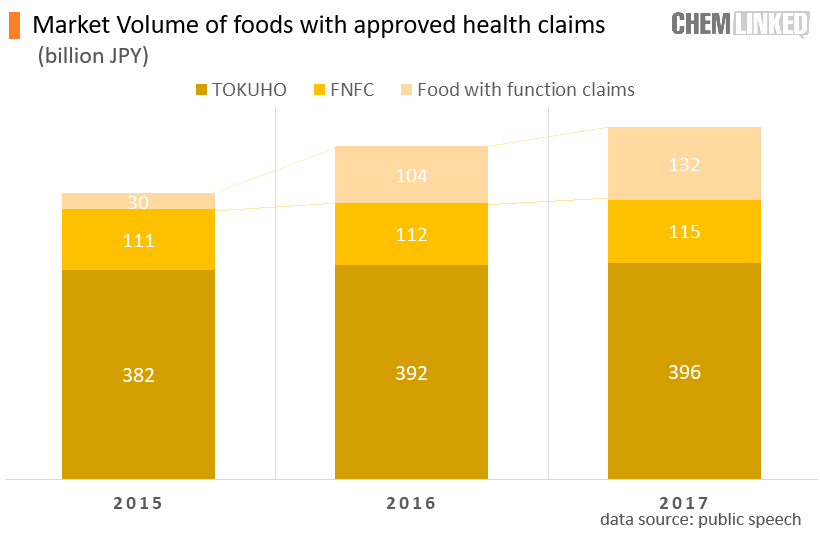
There is another avenue to Japan’s markets for foods with functional claims in which foods can be labeled with function claims based on scientific evidence. Information on the evidence supporting the safety and effectiveness of the product are submitted to the Secretary-General of the Consumer Affairs Agency before the product is marketed. Unlike TOKUHO, the scheme does not require clinical trials. However, business operators are fully responsible for the authenticity and accuracy of the functional claims used as the government does not conduct technical review over foods with function claims. Consumer Affairs Agency simply collects product information submitted by relevant stakeholders and publicize it on a platform to enable consumers to make decision themselves. The certification mark also differs from FOSHU/TOKUHO foods.  This is a relatively new category established in 2015 to stimulate the health industry and offset the negative influences of deflation and slowing growth. As of Jan. 16, 2019, a total of 1714 foods with function claims had been approved. From 2015 to 2017, market volume of food with function claims grew to 132 billion JPY from 30 billion JPY, surpassing that of FNFC. It was projected to top 250 billion JPY in 2018[3].
This is a relatively new category established in 2015 to stimulate the health industry and offset the negative influences of deflation and slowing growth. As of Jan. 16, 2019, a total of 1714 foods with function claims had been approved. From 2015 to 2017, market volume of food with function claims grew to 132 billion JPY from 30 billion JPY, surpassing that of FNFC. It was projected to top 250 billion JPY in 2018[3].
(Data source: Consumer Affairs Agency)
| 2016 | 620 |
| 2017 | 452 |
| 2018 | 621 |
ØJapan VS China
In China, health foods are a category of foods for special dietary purposes which after approval by SAMR can be labelled with both a “blue hat” certification mark and specific functional health claims. Health foods include both foods with functional claims and nutrient supplements. There is no equivalent category to FNFC in China.
Function Claims Adding Value to Yogurt
Yogurt with specified health benefits is one of the most popular health food categories in Japan. Use of active ingredients is a great value multiplier and despite a higher price tag is very attractive to consumers. During the Food and Beverage Innovation Forum 2019 (FBIF 2019) held in Hangzhou last month, keynote speeches offered by delegates from Meiji Holdings Co., Ltd Satoru Ozaki included some data and statistics derived from Japan’s yogurt market. Data showed that most Japanese consumers are conscious of the health benefits of yogurt (see chart below).
ØMeiji Bulgaria yogurt LB81
 This Meiji yogurt is an excellent example of value added yogurt on sale in japan and includes functional claims. It includes the probiotic lactobacillus LB81 and is permitted to use labeling indicating the function “it balances intestinal bacteria and maintains intestinal health by action of lactic acid bacteria LB81.”
This Meiji yogurt is an excellent example of value added yogurt on sale in japan and includes functional claims. It includes the probiotic lactobacillus LB81 and is permitted to use labeling indicating the function “it balances intestinal bacteria and maintains intestinal health by action of lactic acid bacteria LB81.”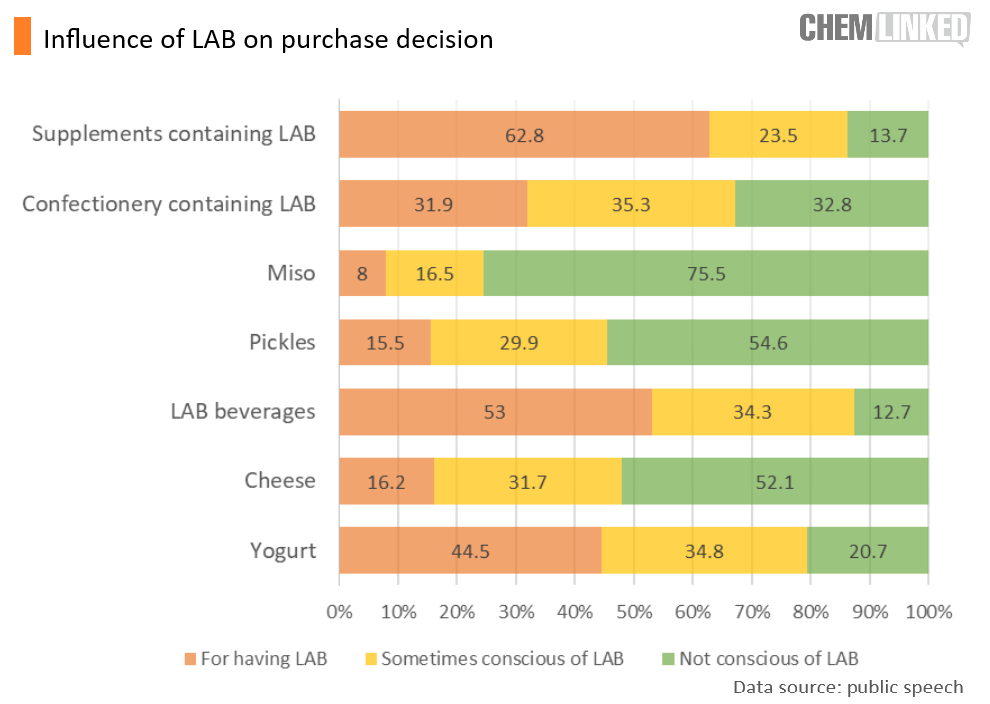 Sales of yogurt account for 37% of Meiji’s revenue, beating its other food lines such as milk, chocolate and whey protein.
Sales of yogurt account for 37% of Meiji’s revenue, beating its other food lines such as milk, chocolate and whey protein.
ØGlico Bifix yogurt
 Another example of a yogurt with function claims is the yogurt shown above which is manufactured by Glico. It was already a popular product before it was marketed as a “food with function claims”. The addition of the labeling claims have helped it gain further market share.
Another example of a yogurt with function claims is the yogurt shown above which is manufactured by Glico. It was already a popular product before it was marketed as a “food with function claims”. The addition of the labeling claims have helped it gain further market share.
Tea with Health Claims
Besides yogurt, another important segment in this market is tea based beverages. “Drinking this product when having a meal could reduce absorption of fat” is a ubiquitous label used on TOKUHO beverages[4]. Common functional claims include reducing sugar and body fat. There are various tea categories, but are all centered on “health”, like black tea, green tea and beverages that mix barley with tea.
Thanks to increasing consumer health awareness and Japan’s deeply entrenched tea culture, TOKUHO beverages are performing extremely well and now major multinationals like Suntory, Kirin, Coca-Cola and even Kao (a chemical and cosmetics company headquartered in Tokyo) are competing for market share.
ØSuntory Iyemon Tokucha
According to daily product ranking[5] updated on a leading ecommerce platform Rakuten, 7 out of the top 10 best-selling TOKUHO sold on the platform are tea beverages. A Suntory green tea drink occupies the number 1 slot. 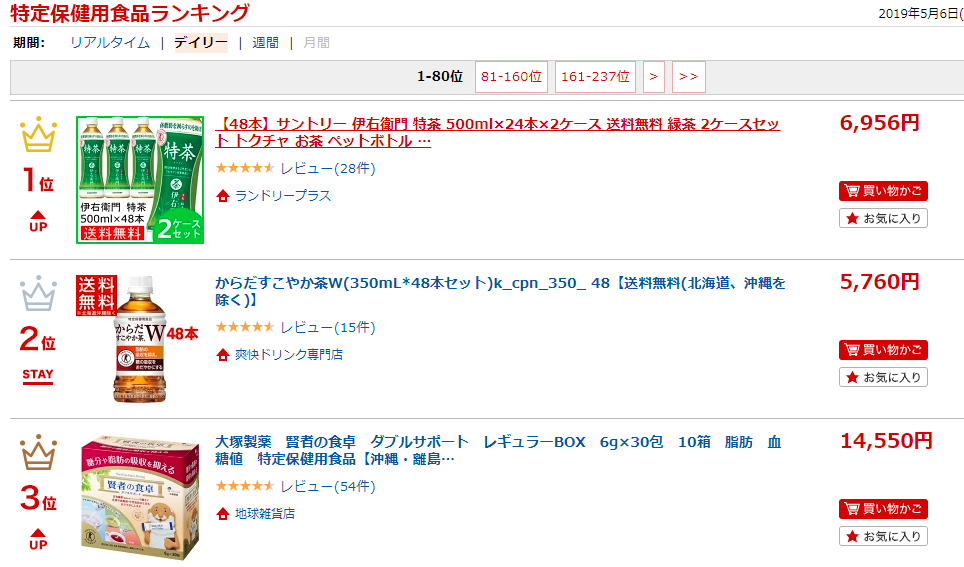 According to research carried out earlier in 2015 by Suntory, 43.8% of respondents had drank TOKUHO beverages, 34.2% drank one a week. Most respondents cited their reasons for purchasing as “it is good for my health” or “I don’t want to gain weight after eating fatty foods.”
According to research carried out earlier in 2015 by Suntory, 43.8% of respondents had drank TOKUHO beverages, 34.2% drank one a week. Most respondents cited their reasons for purchasing as “it is good for my health” or “I don’t want to gain weight after eating fatty foods.”
Read More


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










