On April 30, 2019, NMPA (National Medical Products Administration) issued a notice detailing 21 newly registered infant formula products. An additional 3 registration applications were rejected as revealed by NMPA notice (approved [1], rejected [2]).
Analysis of the 24 infant formula registration applications
This update includes 7 product lines (1 production line for goats milk infant formula), amounting to 21 infant formula products from 3 Chinese enterprises.
| Country | Enterprises | Products | |
| Approved | China | Canobank | 3 production lines, 9 products |
| Hunan Zhanhui Food | 3 production lines, 9 products (including 1 production line for infant formula goat milk) | ||
| Junlebao | 1 production line, 3 products | ||
| Rejected | New Zealand | Dairy Nutraceuticals Limited | 1 production line for infant formula goat milk, 3 products |
A brief introduction to the four enterprises
Canobank [3] was founded in October 2006 and was formerly an affiliate of Conba Group. It is now dedicated to the research & development, production and sales of mother care and baby products.
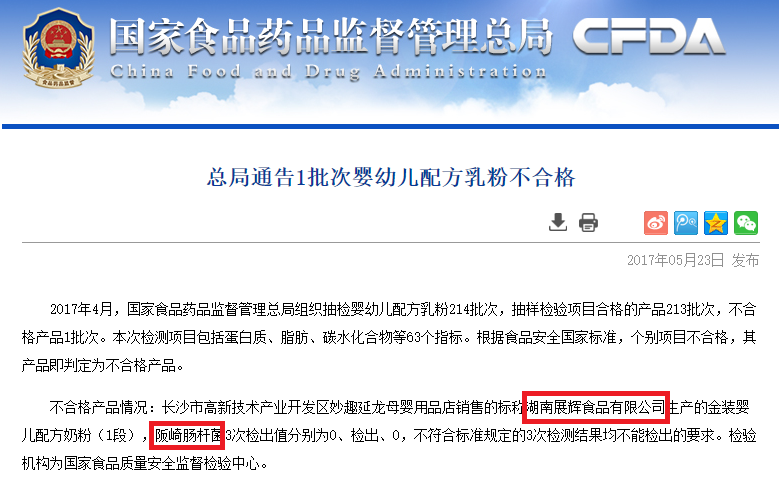
Hunan Zhanhui Food [4] blacklisted by CFDA twice in 2017. On May 23rd, 2017 [5], one of its products tested positive for Enterobacter sakazakii. On August 30th, 2017 [6], 31 items were found noncompliant during an inspection of the company’s food safety production system.
Junlebao [7]:
According to Liu Senmiao, the vice president of Junlebao, in 2018, the sales volume of Junlebao’s infant formula products topped 46 thousand tons, with a sales income of over 5 billion yuan. The YOY growth in sales was in excess of 100%. Until now, Junlebao has five production lines in two factories.
Dairy Nutraceuticals Limited [8] has passed the CNCA registration for dairy products but has still not registered its products.
Status Quo
Currently, 162 enterprises 1253 formulae (excluding special infant formula) have won China's approval, check all in ChemLinked's flist [9] (the infant formulae registered this time are excluded, for the authority hasn't updated the info of these products on its database). Goat milk powder accounts for 21% of total registrations. 50 overseas manufacturers have completed the registration of 304 formulae, representing 24.3% of all infant formula registered in China.


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










