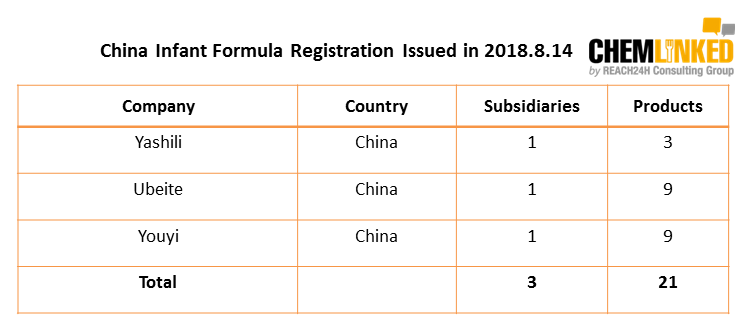
On Aug. 14th, 2018, China SAMR unveiled the latest infant formula registration list, including 21 products from 3 domestic enterprises.
According to SAMR’s “3 stipulation scheme”, a “special food safety supervision department” has been set up to undertake the exclusive management of special food issues. Therefore, we can expect the frequency of approval announcements regarding infant formula. Besides, SAMR will attach greater importance to the in-process and post-market supervision of infant formula in future.
Currently, 153 enterprises 1177 formulae (excluding special infant formula) have won China’s approval (check all in our F-lists). Goat milk powder accounts for 22% of total registrations. 47 overseas manufacturers have completed the registration of 280 formulae, representing 24% of all infant formula registered in China.
Top 5 overseas registrants who have their products granted registration approval
Group | Product number | Brands number |
Synutra | 27 | 9 |
Nestle | 25 | 10 |
| Abbott | 21 | 8 |
Danone | 21 | 6 |
Health & Happiness Group (H&H Group) | 15 | 5 |
Here are the previous batches of registered infant formula
|
|
|
|
|
|


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










