On November 9, 2022, the National Pharmaceutical Regulatory Agency (NPRA) of the Ministry of Health Malaysia issued a “Guidance on the Acceptance Criteria of Quantification by Input (QBI) of Active Ingredients Claimed on Label of Traditional Medicine and Health Supplement (TMHS) Products1 (hereinafter the Guidance) to expound the situations in which the active ingredients in TMHS products could be quantified by input.
Supervision of Health Supplement in Malaysia
In Malaysia, regular foods are supervised by the Food Safety and Quality Division (FSQD) of the Ministry of Health, and drugs are regulated by NPRA. When it comes to “nutraceuticals” or the so-called health products—generally, these products are a combination of food ingredients and active ingredients for oral consumption—sometimes it is difficult to define whether they are foods or drugs. Such products are termed as “food-drug interphase (FDI) products” by Malaysian authority.
In 2000, to better define and regulate such products, FSQD and NPRA jointly formed the Committee for the Classification of Food-Drug Interphase Products. The authority has given a pictorial guide (as below) to help stakeholders determine the classification between food and drug. Applicant may also verify FDI product classification by seeking classification service from NPRA2.
Pictorial Guide for Classification of Food or Drug
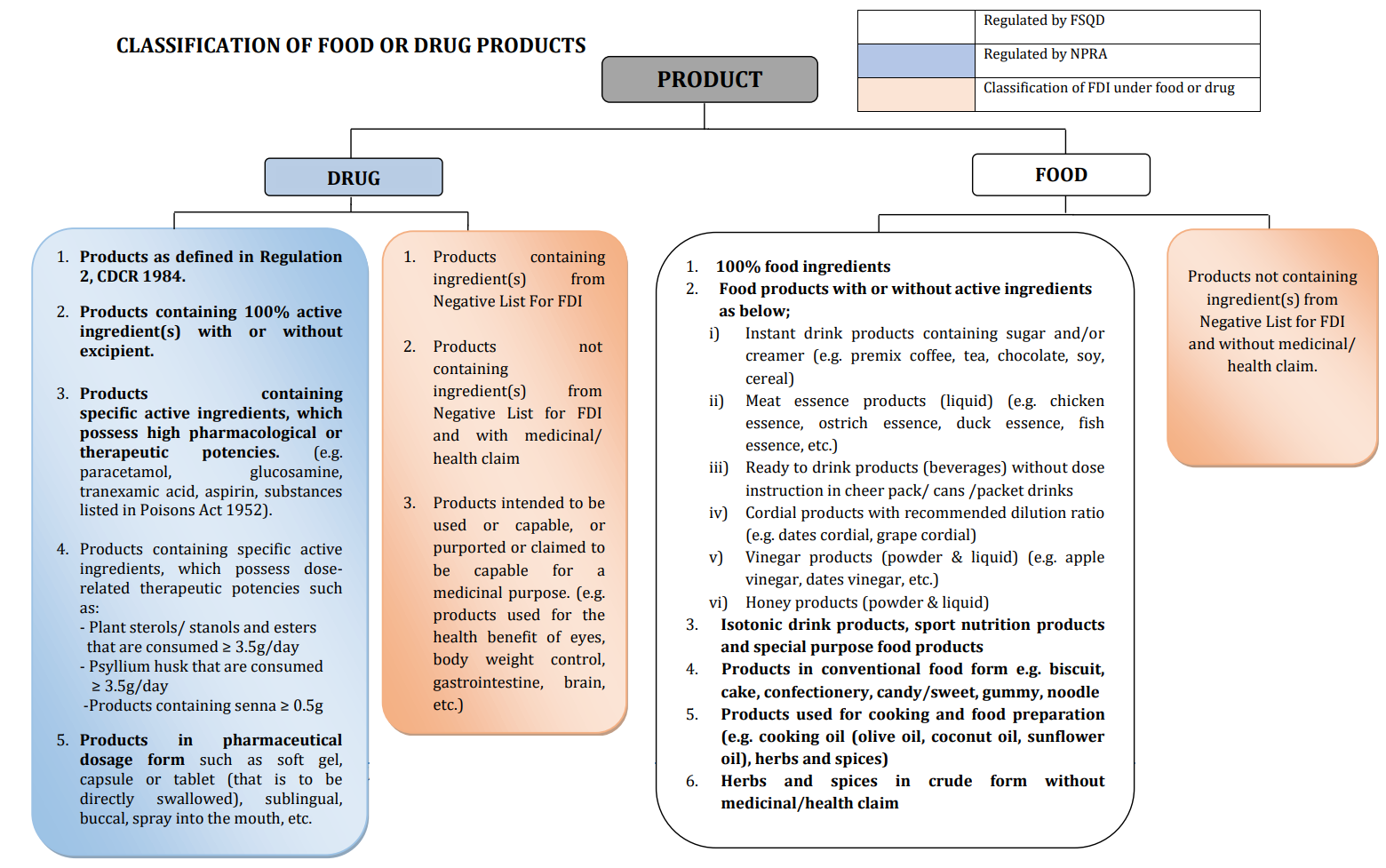
Source: Drug Registration Guidance Document (DRGD) Third Edition, Third Revision July 20223
Malaysia does have a product type called “health supplement” at the regulatory level. According to the regulation, health supplement refers to any product used to supplement a diet and to maintain, enhance as well as improve the health function of human body. It is presented in small unit dosage forms (to be administered) such as capsules, tablets, powder, liquids and shall not include any sterile preparations (i.e. injectables, eyedrops). It may contain one or more of the following combinations:
Vitamins, minerals, amino acids, fatty acids, enzymes, probiotics, and other bioactive substances;
Substances derived from natural sources, including animal, mineral and botanical materials in the forms of extracts, isolates, concentrates, or metabolite;
Synthetic sources of ingredients mentioned in 1 and 2, which may only be used where the safety of these has been proven.
After comparing the definition of health supplement to the “Pictorial Guide for Classification of Food or Drug” above, it is clear that health supplement is regulated as drugs/medicinal products in Malaysia. As per the Drug Registration Guidance Document (DRGD) Third Edition, Third Revision July 2022, medicinal products (including health supplements and natural products like herbs) shall get registered with the authority.
The Introduction of QBI Method
During the registration of health supplement (please refer to Appendix 6 of Malaysian Drug Registration Guidance Document: Guideline on Registration of Health Supplements), it is required to conduct quantitative assay test for all active ingredients claimed on the product label. However, this may not be practical to achieve in some cases. Therefore, the authority introduces the QBI method. The definition of QBI is borrowed from the Australian Therapeutic Goods Administration (TGA)—“QBI means the content of an active ingredient is estimated from the amount dispensed during the manufacture of the product. This practice is done when the assay of active ingredient could not be achieved in the finished product.”
To summarize, QBI is allowed when it meets the following criteria:
There is no assay method for active ingredients in the monograph/compendial (pharmacopoeia)
There is a justification that can be supported with scientific references and test report showing the test results of the material cannot be achieved due to the quantity of active ingredients
If QBI is allowed, the finished product’s manufacturer must conduct an assay on the active ingredient raw material.
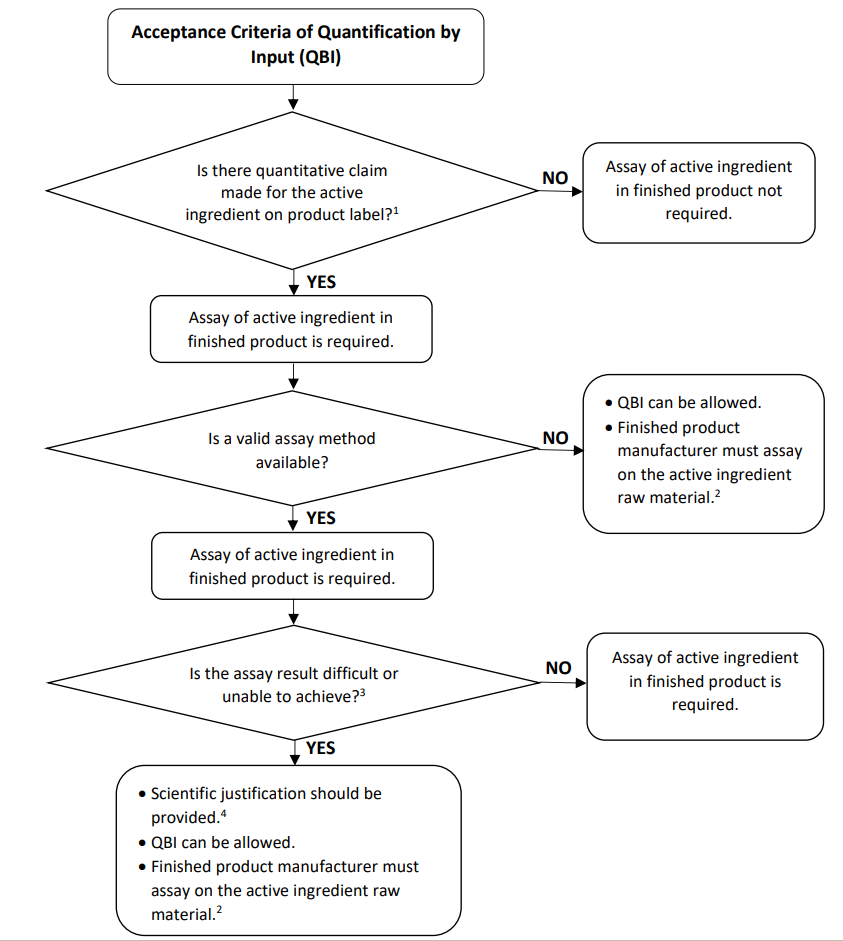
Determination of QBI acceptance criteria is illustrated in the flow chart below. Details about the acceptance criteria for QBI and examples of QBI calculation can be found in the Guidance. The implementation of the Guidance will enter into effect in January 2023.
Flow Chart: Acceptance Criteria of Quantification by Input (QBI) of Active Ingredients Claimed on Label of Traditional Medicine and Health Supplement (TMHS) Products


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










