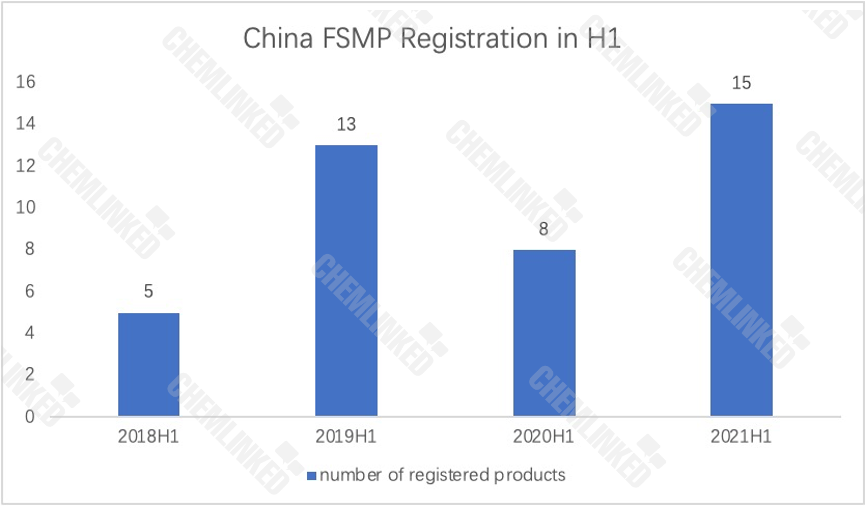
FSMP registration in 2021H1
As revealed by SAMR, in the first half of 2021, 15 food for special medical purposes products were granted registration approval. All of them are domestic brands.
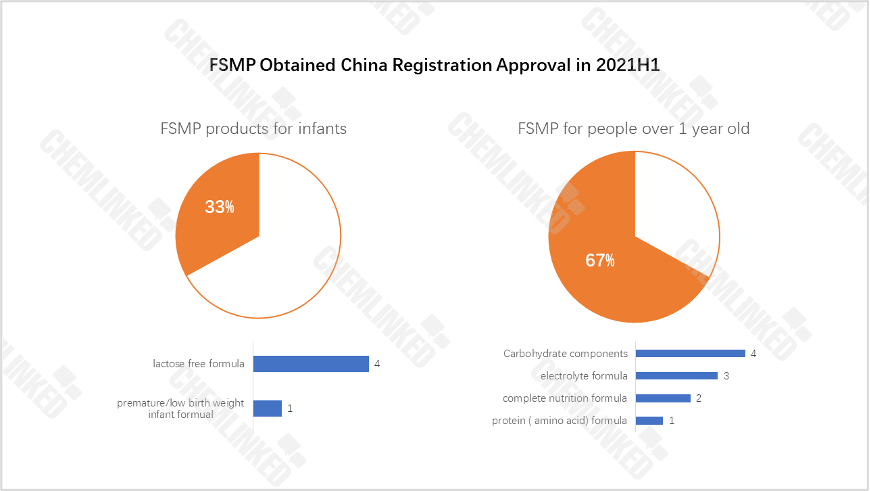
FSMP could be classified into products designed for infants (below one year old) and products targeted at people over 1 years old. The data below showcased that the latter accounted for the majority of FSMP obtained approval in 2021H1. In terms of product type, carbohydrate components occupied the most. As for FSMP for infants, lactose free formula is the type that has more approval.
FSMP registration by 2021H1
By June 30, 2021, SAMR has announced the registration approval of 72 FSMP products, among which domestic products accounts for around 60% (check all products in Flist). For imported products, Netherlands has most registered FSMP (8), followed by UK (4) and Germany (3).
 When it comes to the product types, FSMP for infants and for people over 1 years old both occupy 50% of registered FSMP by 2021H1. The number of premature/low birth weight infant formula (12) ranks the top of all infant FSMP, followed by lactose free formula (10) and partially-hydrolyzed lactoprotein infant formula (6). In terms of FSMP targeted at people over 1 years old, complete nutrition formula occupies the most (19).
When it comes to the product types, FSMP for infants and for people over 1 years old both occupy 50% of registered FSMP by 2021H1. The number of premature/low birth weight infant formula (12) ranks the top of all infant FSMP, followed by lactose free formula (10) and partially-hydrolyzed lactoprotein infant formula (6). In terms of FSMP targeted at people over 1 years old, complete nutrition formula occupies the most (19).
Product Type | Number | ||
FSMP for over 1-year-old People (36) | Nutritionally Complete Formula Food (19) | Nutritionally complete formula food | 19 |
Nutritionally Incomplete Formula Food (17) | Electrolyte formula | 6 | |
Carbohydrate components | 5 | ||
Protein (amino acid) components | 4 | ||
Formula for amino acid metabolic patients | 2 | ||
Specific Full Nutritional Formula Food | / | 0 | |
FSMP for Infants (36) | / | Premature or low birth weight infant formula | 12 |
Lactose free formula | 10 | ||
Partially hydrolyzed milk protein formula | 6 | ||
Extensively hydrolyzed milk protein formula | 4 | ||
Infant nutrition supplement | 3 | ||
Amino acid metabolism disorder formula | 1 | ||
Up to now, 30 domestic and overseas enterprises have their FSMP products registered in China, among which Nestlé occupies the most.
Group/Enterprises | Product Number |
Nestlé | 11 |
ABBOTT | 8 |
Hengrui Jian Kang | 5 |
Synutra | 5 |
Beingmate | 4 |
SHS | 4 |
Mead Johnson | 3 |
Junjoy | 3 |
M.F.nutrition | 3 |
AusNuotore | 2 |
Yipin | 2 |
Duoxin | 2 |
Zhengda fenghai | 2 |
Maeil Dairies | 2 |
Donglianchangfu | 1 |
Daisy FSMP | 1 |
Ballansat | 1 |
Hisun (group) | 1 |
Aumix | 1 |
Wissun | 1 |
Ausnutria | 1 |
Wyeth | 1 |
EnterNutr | 1 |
Yili | 1 |
Nantong Richen | 1 |
Yabao | 1 |
Firmus | 1 |
Aisheng | 1 |
Milupa Gmbh | 1 |
Kwins Health | 1 |
Trend of FSMP registration
1. Optimized Regulation Brings Better Regulatory Environment
“Administrative Measures for Registration of Foods for Special Medical Purpose” issued in 2016 specified the requirements, application procedure and supervision of FSMP registration. Years ago the competent authority has internally embarked upon the amendment of the Measures and the revised version was included in SAMR legislation scheme of 2021. It is said that the FSMP products will be subject to higher technique standard and more stringent supervision requirements. Meanwhile, some administrative procedures will be simplified for efficiency. For example, the alteration of manufacturers’names and their addresses will be verified and approved onsite (read ChemLinked news for more details). However, it still needs some time for the promulgation of the new version, because the revision to the Measures was listed as type 2 legislation item, which means the competent authority will promote the legislative progress efficiently, but no specific deadline is given.
2. Innovative onsite inspection helps to remove product registration obstacles
Onsite inspection is usually the last step to obtain the product registration approval. However, the schedule of onsite inspection is always uncertain for overseas manufacturers. In addition, affected by COVID-19, it seems impossible for Chinese experts to review overseas factories in recent years. In this case, China is seeking for innovative methods to conduct the onsite inspection. At the end of June 2021, China announced to solicit public opinions on the methods for onsite inspection of infant formula recipe and FSMP product registration. There are four ways under discussion: 1) cooperation with overseas competent authorities by signing memorandums; 2) Video inspection; 3) Third-party institute inspection; 4) Self-inspection by enterprises. So far it still remains unknown that what measures will be taken, but it showcases the competent authority is making efforts to remove the obstacles for FSMP registration.
Notes:
Data are sourced from Special Food Information Query Platform. As the database does not provide a real-time update, there may be inconsistency with the fact.


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










