FSMP registration during 2020 H1
According to SAMR data, eight food for special medical purposes products (FSMP) obtained China’s registration approval during the first half of 2020. Seven of them are domestic products and the other is imported from the Netherlands. Nutritionally complete formula accounts for the majority of the overall approval in 2020 H1, representing 75%.
S/N | Enterprise | Product name | Registration No. | Country |
1 | 爱优诺营养品有限公司 | 爱优诺优益力 nutritionally complete formula | 国食注字TY20200001 | China |
2 | 爱优诺营养品有限公司 | 爱优诺优康力 nutritionally complete formula | 国食注字TY20200002 | China |
3 | 广东君悦营养医学有限公司 | 君蓓全 nutritionally complete formula | 国食注字TY20200004 | China |
4 | 哈尔滨拜仑斯特临床营养有限公司 | 唯卡能® nutritionally complete formula | 国食注字TY20200003 | China |
5 | 苏州恒瑞健康科技有限公司 | 希瑞臻 nutritionally complete formula | 国食注字TY20200005 | China |
6 | 杭州贝因美母婴营养品有限公司 | 舒力乐 partially-hydrolyzed lactoprotein infant formula | 国食注字TY20200006 | China |
7 | 亚宝药业集团股份有限公司 | 唯源素® nutritionally complete formula | 国食注字TY20200007 | China |
8 | Nestle Nederland B.V. | 恩敏舒 amino acid-based infant formula | 国食注字TY20205001 | the Netherlands |
The amount of FSMP registration approval declined compared with the same time last year. The outbreak of COVID-19 at the beginning of 2020 is deemed as the major factor that postponed the special food registration (click here for the details).
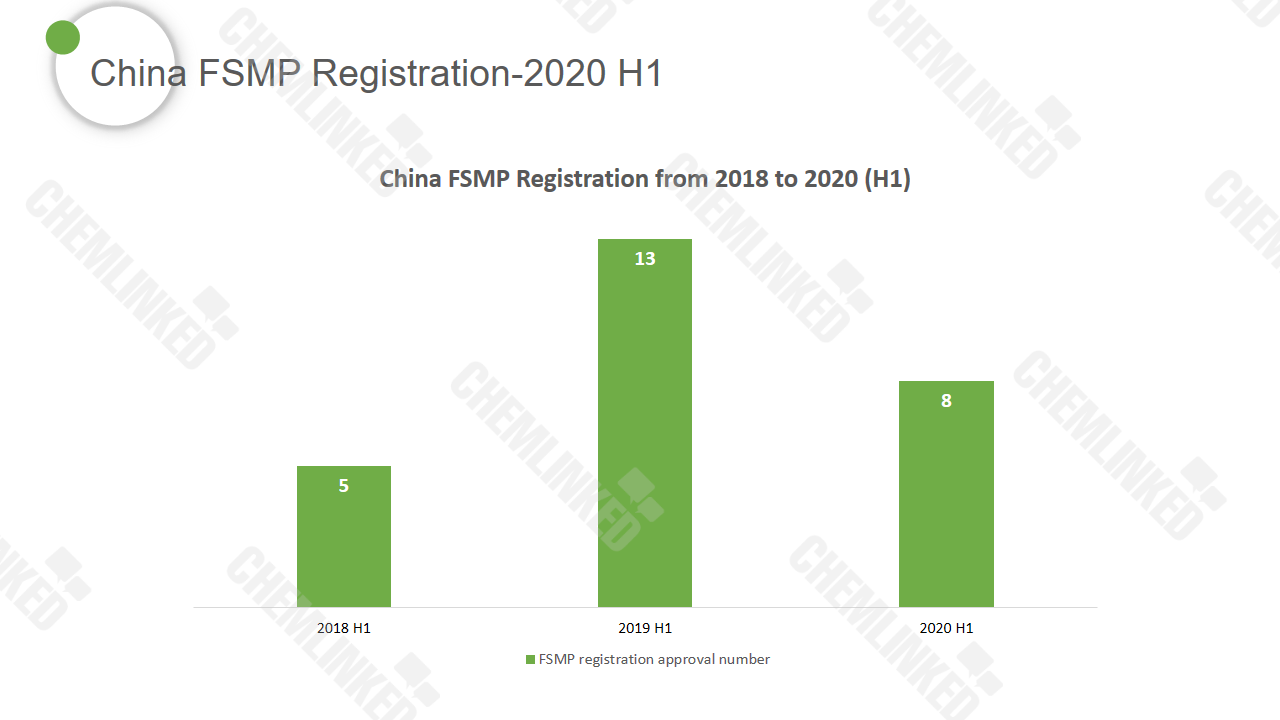
It is noteworthy that four products failed the registration this year (all domestic products), which are:
伊正佳™ nutritionally complete formula
赛速康nutritionally incomplete formula food (electrolyte formula food)
沭泰electrolyte formula food
美德能舒闲宁® nutritionally complete formula food
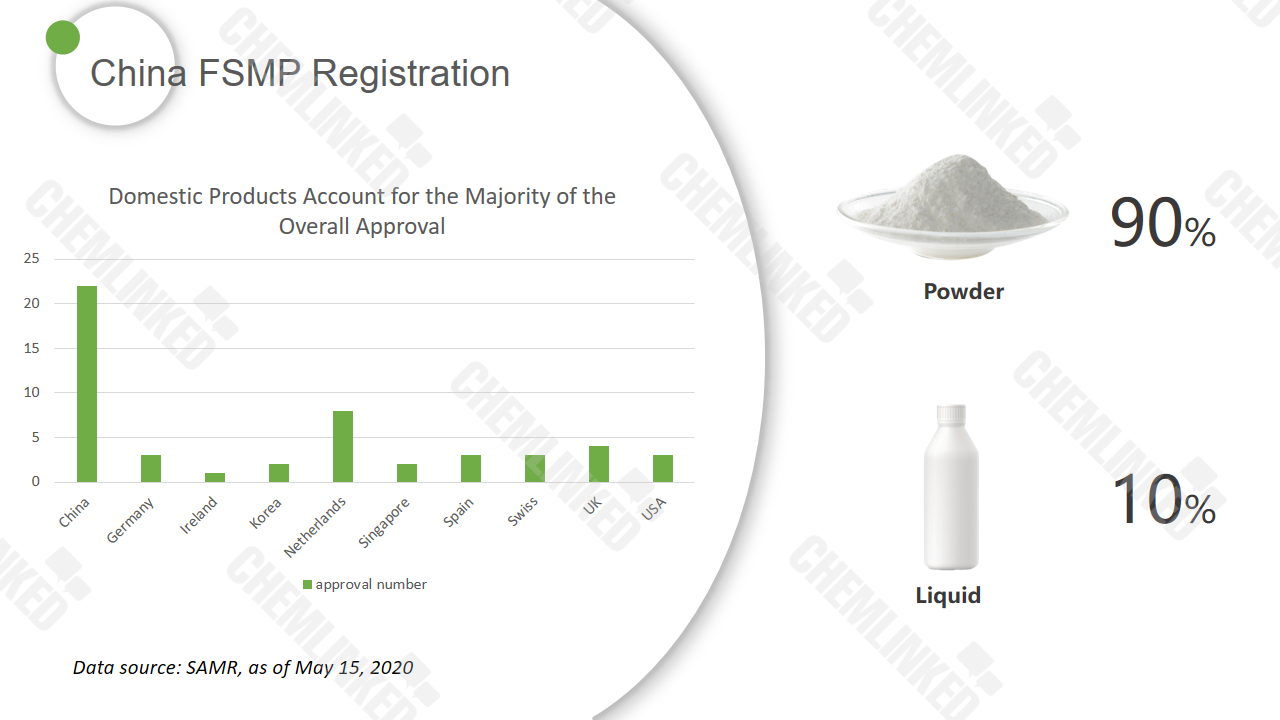
The overview of FSMP registration
As of May 15, 2020, China has granted registration approval of 51 FSMP products, and domestic products account for 43% of the total registration. According to the product information unveiled by SAMR, 90% of those approved products are in powder form.
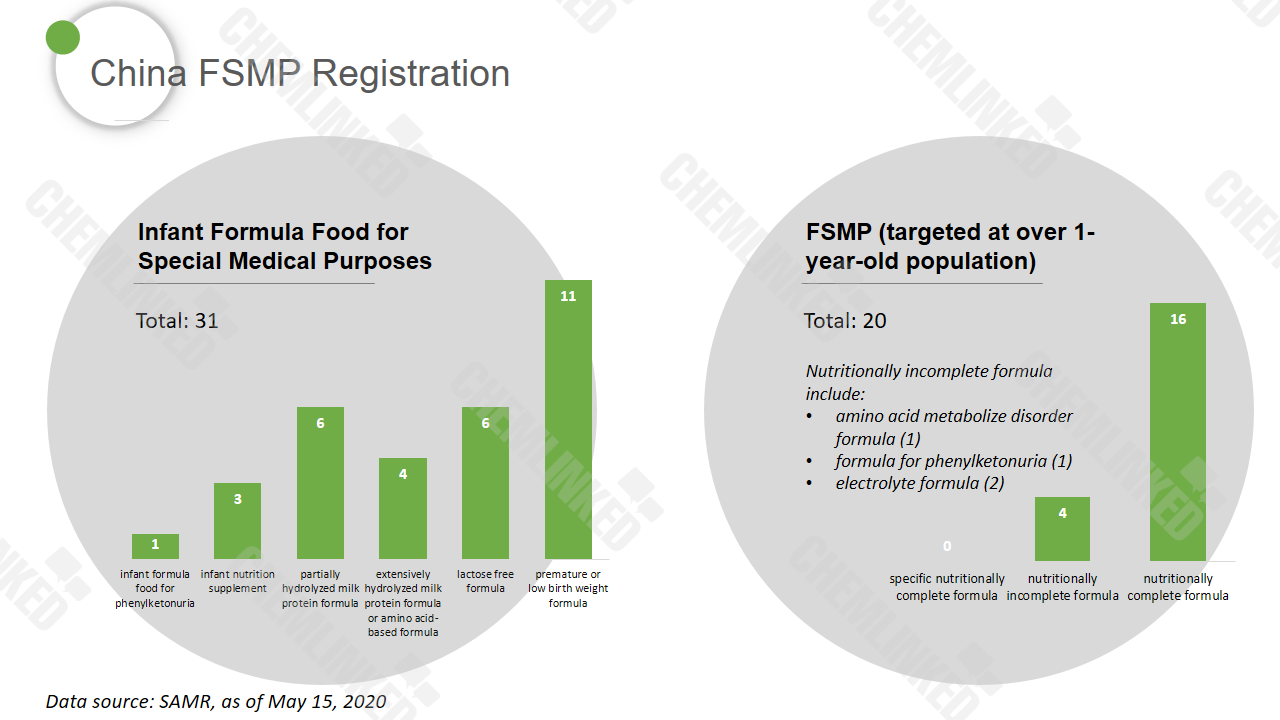
 In terms of the product type, infant formula food for special medical purposes represents 60.8%, and quite a few of them are infant premature or low birth weight formula. FSMP targeting at the population over 1-year-old could be classified as nutritionally complete formula, nutritionally incomplete formula food and specific nutritionally complete formula. Currently, none specific nutritionally complete formula product has won approval.
In terms of the product type, infant formula food for special medical purposes represents 60.8%, and quite a few of them are infant premature or low birth weight formula. FSMP targeting at the population over 1-year-old could be classified as nutritionally complete formula, nutritionally incomplete formula food and specific nutritionally complete formula. Currently, none specific nutritionally complete formula product has won approval.
 Nestlé group obtained the most FSMP registration certificates in China, amounting to 11 products.
Nestlé group obtained the most FSMP registration certificates in China, amounting to 11 products.
Enterprise | Approval amount |
Nestlé (Group) | 11 |
ABBOTT (Group) | 8 |
Synutra Nutrition Food Co., Ltd. | 5 |
Shs International Ltd | 4 |
Hangzhou Beingmate Maternal And Infant Nutrition Food Co., Ltd. | 4 |
Mead Johnson Netherlands Co., Ltd | 3 |
Suzhou Hengrui Jian Kang Technology Co., Ltd. | 3 |
Aiyounuo Nutrition Food Co., Ltd. | 2 |
Pyeongtaek Factory, Maeil Dairies Co.,Ltd. | 2 |
Milupa Gmbh | 1 |
Wyeth Nutritionals Ireland Ltd. | 1 |
Dorbet Yili Dairy Co., Ltd | 1 |
Guangdong Junjoy Medical Nutrition Co., Ltd. | 1 |
Harbin Ballansat Clinical Nutrition Co., Ltd | 1 |
Jilin M.F. Nutrition Technology Co., Ltd. Changchun Branch | 1 |
Nantong Richen Bioengineering Co., Ltd. | 1 |
1 | |
Yabao Pharmaceutical Group Co., Ltd. | 1 |
It may be harder to get FSMP registration certificate in 2020
From my personal perspective, the amount of FSMP registration approval in 2020 may not exceed that in the last year. On the one hand, the COVID-19 has somewhat postponed the technical review of the competent authority in the first half year, and the pandemic may also affect the registration schedule of some companies.
In May this year, infant related food safety crsis happened again in China, which is associated with the consumption of a solid beverage marketed and sold as an FSMP (click here for the details). Later on, the competent authority at all levels immediately started the specialized campaign to crack down on false advertising and illegal sales. In this case, SAMR may place more emphasis on the market supervision of FSMP in this year.
Additionally, China consulted on Special Food Registration Onsite Inspection Requirements in July 2019. If China passes the consultation draft this year, it will be harder to get the product approval since onsite inspection will become a mandatory step.


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










