On Dec. 2, 2019, State Administration for Market Regulation (SAMR) released consultation drafts of the Directory of Raw Materials Used in Nutrient Supplementary Health Food (2019 Edition) 《营养素补充剂保健食品原料目录(征求意见稿)(2019年版)》 and the Directory of Functional Claims Allowed in Nutrient Supplementary Health Food(2019 Edition) 《允许营养素补充剂保健食品声称的保健功能目录(征求意见稿)(2019年版). The drafts are currently undergoing public consultation.[1]
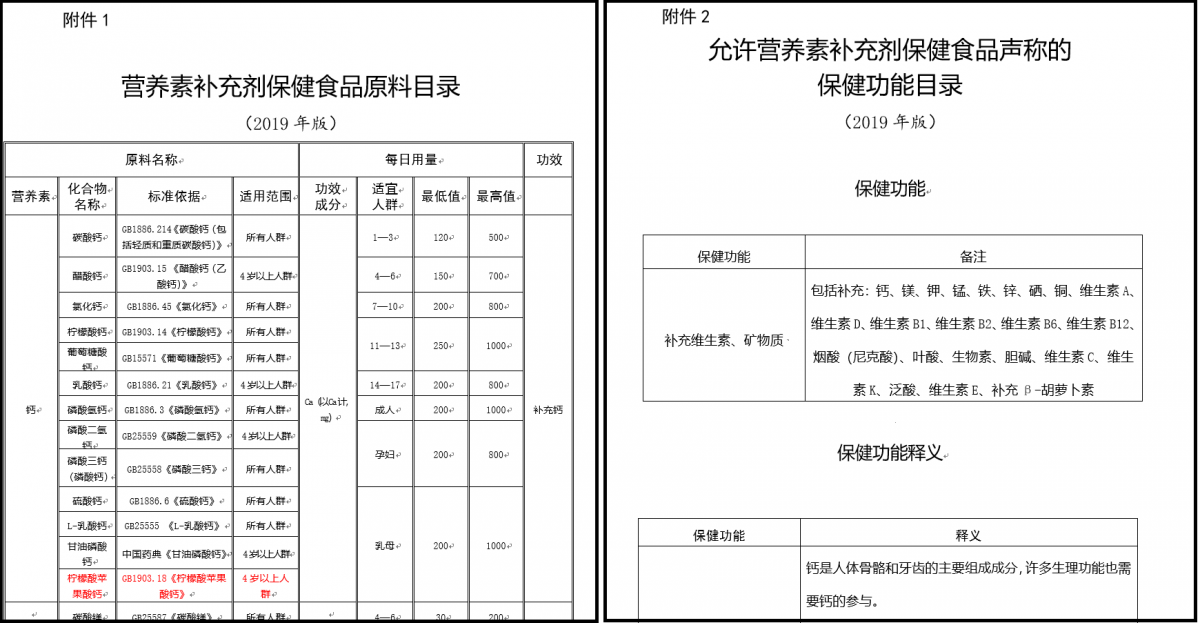 The two drafts are amendments to Health Food Raw Materials Directory (1st batch)[2] and Health Food Functional Claims Directory (1st batch)[3] which were published in 2016. Despite changes to the document name, significant changes are:
The two drafts are amendments to Health Food Raw Materials Directory (1st batch)[2] and Health Food Functional Claims Directory (1st batch)[3] which were published in 2016. Despite changes to the document name, significant changes are:
Inclusion of New Raw Materials
Newly added nutrient sources
| Nutrient | Newly added ingredient | Standard of the ingredient | Target group |
| Calcium (Ca) | Calcium citrate malate | GB 1903.18 | People aged above 4 |
| Magnesium (Mg) | Magnesium gluconate | GB 1903.29 | All people |
| Iron (Fe) | ferric pyrophosphate | GB 1903.16 | All people |
| ferric citrate | GB 1903.37 | All people | |
| Zinc (Zn) | Zinc acetate | GB 1903.35 | All people |
| Choline | Choline chloride | GB 1903.36 | All people |
Newly added nutrient and claim
| Newly added nutrient | Standards of the ingredient | Target group | Newly added claim |
| β-carotene | GB 8821; GB 28310; GB 31624 | Grown-up | Supplementβ-carotene |
In the current directory of raw materials, β-carotene is one of the permitted sources of Vitamin A that can be used in Vit A supplements. In the new draft it is specified that “blue hat” certified nutrient supplements containing this can use the claim "supplementing β-carotene”.
Nutrient Claims Stipulated in GB 28050 Might Extend to Blue-hat Nutrient Supplements
According to the draft of the Directory of Functional Claims Allowed in Nutrient Supplementary Health Food (2019 Edition), health food nutrient supplements will also be allowed to use the nutrient function claims specified in GB28050.
| Health claim | Additional nutrient function claims |
| Supplement Calcium |
|
| Supplement Iron |
|
| Supplement Vitamin D |
|
| Supplement Vitamin B2 |
|
| For the full list, please refer to the consultation draft. | |
These nutrient function claims are laid out in GB 28050 National Food Safety Standard General Rules for Nutrition Labeling of Prepackaged Foods but are inapplicable to health food and special dietary food.[4]During the 3rd China Health Food Conference held in Zhuhai, SAMR official Wan Chao hinted that proposed changes in the draft regulation had been made to address disparities between the broad scope of nutrient function claims permitted on general foods, and limited range of nutrient supplementation claims allowed to be labeled on health food nutrient supplements. Currently, "Blue Hat" certified nutrient supplements are limited to nutrient supplementation claims like “supplements Calcium intake" despite having relatively stringent regulatory compliance demands. Somewhat counterintuitively fortified general foods can use a much broader scope of nutrient function, and content claims (specified in table above) such as "Vitamin E has antioxidant properties" despite having reduced compliance demands compared to nutrient supplements. (read previous regulatory analysis: Capitalizing on General Food Nutrient Function Claims)[5]
Download the drafts here:
Directory of Raw Materials Used in Nutrient Supplementary Health Food (2019 Edition)
Directory of Functional Claims Allowed in Nutrient Supplementary Health Food (2019 Edition)
Any comments shall be submitted before December 31, 2019, via one of the following approaches:
SAMR official website: http://www.samr.gov.cn
Email: [email protected]
Mail: Special Food Agency of SAMR, No. 1 Beilu Garden, Xicheng District, Beijing, China.


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










