Content of the notification goes as follows:
1. Applicable entities
Qualified and accredited enterprises that produce and sell infant formula or health food in countries of origin.
2. Applicable products
1) imported health foods that should undergo registration or filing but haven’t gained the certificate of registration or filing yet.
2) imported infant formula that haven’t gained certificate of recipe registration issued by SAMR.
3. Procedure of Application for Temporary License
1)Temporary registration of infant formula/health food
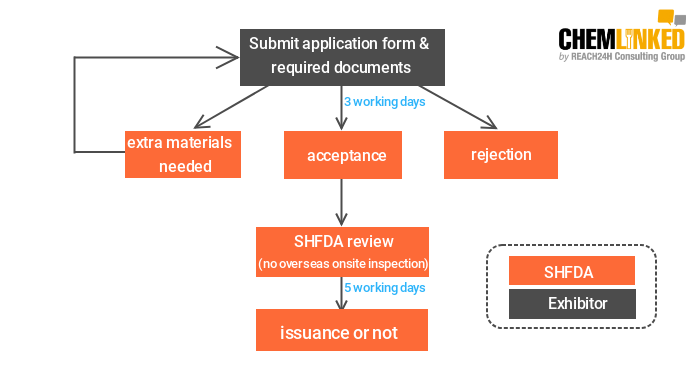
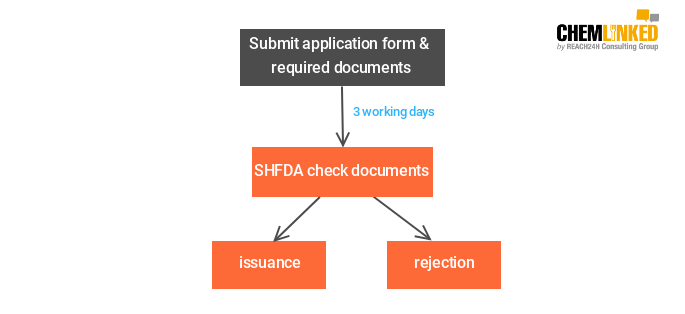
2)Temporary filing of health food
*Download application form
Annex 1: application form of infant formula temporary registration
Annex 2: application form of health food temporary registration
Annex 3: application form of health food temporary filing
Other
Temporary license is valid only during CIIE and within the exhibition.
Infant formula or health food granted temporary license are not required to display their temporary registration/filing number on product packages.
Address: Shanghai Food and Drug Administration service center (tel: 021-23111111)


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










