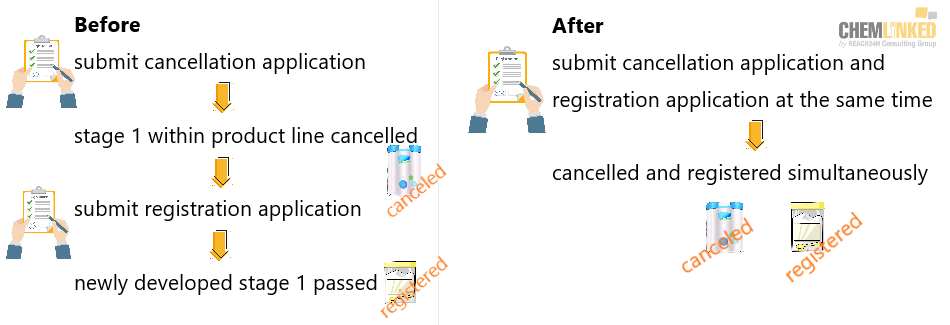
On Jan. 8, 2019, China began allowing the simultaneous cancellation and replacement of a previously registered infant formula product line stage e.g. cancellation and replacement of a stage 1 product within a product line with a newly registered stage 1.
As we know for general trade channels, China limits each GAC approved infant formula manufacturing facility to placement of 3 infant formula product lines on the market (each product line having 3 stages designed to meet the specific nutritional requirements of the growing baby/toddler/child.)
Previously, if a manufacturer decided to remove one stage of a product line and place a newly approved registered infant formula product on the market there was a considerable delay between cancellation and approval. During that lag period a GAC approved manufacturer would sometimes be limited to marketing 8 products.
The new system offers the following benefits:
No time gap: date of cancellation is the beginning of new registration certificate
More than 9 products circulated in the market: old infant formula products produced before cancellation date & new infant formula
Facilitates inventory management and supply/demand refinement


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










