On Sept. 13th[1], Japan's Ministry of Health, Labor and Welfare announced that from October 1st, the sales of agricultural water products using gene-editing technology will be accepted subject to prior review and approval by authorities.
Currently, Japan is in the process of commercializing snapper genetically edited be larger and tomatoes genetically edited to contain a higher concentration of a blood pressure-lowering bioactive substance, which, if approved, could be sold as food in Japan this year.
To learn more about gene-modified organism (GMO) and gene editing please click here.
Later on on Sep. 19, 2019, No.4 announcement issued by MHLW [2] detailed the administrative measures for managing gene-edited foods which are shown below [3]:
1. Prior consultation will determine whether registration is enough or whether a safety review is necessary. For most gene-edited foods, there is no need to conduct a safety review, they are only required to be registered before sale, and filing is applicable to products satisfying one of the following conditions:
The food product is obtained through gene-edit technology
The food product is produced through gene-edited microorganism
2. There is no need to carry out strict safety review for foods that only delete/silence/reduce/amplify specific gene functions without adding external/extra genes. Most of the varieties currently being developed fall into this category. Before these foods can be marketed, developers or importers only need to register the method of gene editing, and whether new allergens have been created. The registration content will be published on the home page of the Ministry of Health, Labor and Welfare [4].
Earlier on Sept. 13th, Japan convened an expert meeting to solicit opinions on this policy. At that time, there was a lot of talk about requiring gene-edited foods to undergo safety review, but the ministry government insisted that the risks could be controlled according to the management measures they developed.
Here is the procedure for the administrative approval of gene-edited food: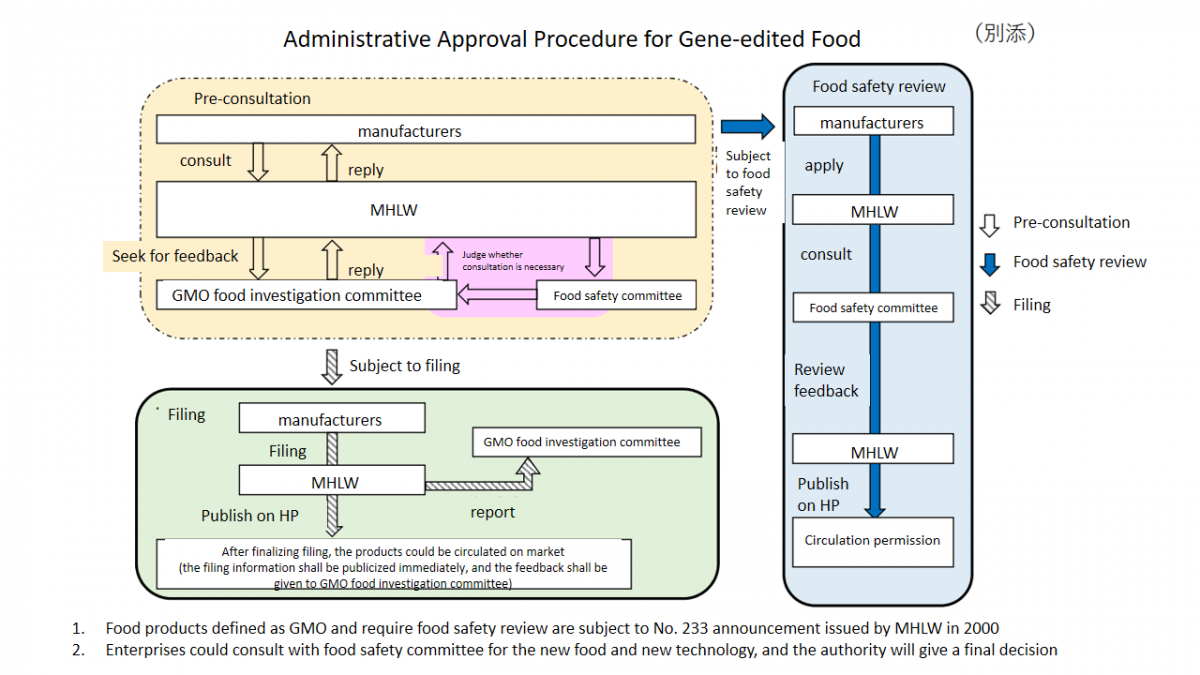
On the same day, MHLW issued the 0919 No.3 announcement [5], which details application requirements for gene-edited food filing and stated that the application document shall include:
Product type and introduction (usage method and purpose)
Gene-edit technology and characteristics that been modified
Confirmation that there is no foreign gene or its residue
Confirmation that the DNA changes do not have negative impact on human health
Nutrient changes after gene editing
The proposed retail date
In addition, the Japanese government is discussing rules on food labelling of gene-edited food. Although Japan’s consumer affairs agency has outlined the difficulties of imposing a mandatory labelling requirement, Yasunori Yoshida, the supervisor of the Ministry food standards review division, points out: "labelling instructions will be included so that the public can properly understand what gene-edited food is and what the risks are."
To read more about the administration of gene-edited food and labelling in Japan please click here.


 Request a Demo
Request a Demo
 We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global food market entry services (including product registration, ingredient review, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










